Search Thermo Fisher Scientific
The Wohl–Ziegler reaction, or the Wohl–Ziegler bromination, is a type of the free radical substitution

The Wohl–Ziegler reaction is a chemical reaction that involves the allylic or benzylic bromination of hydrocarbons using an N-bromosuccinimide and a radical initiator.
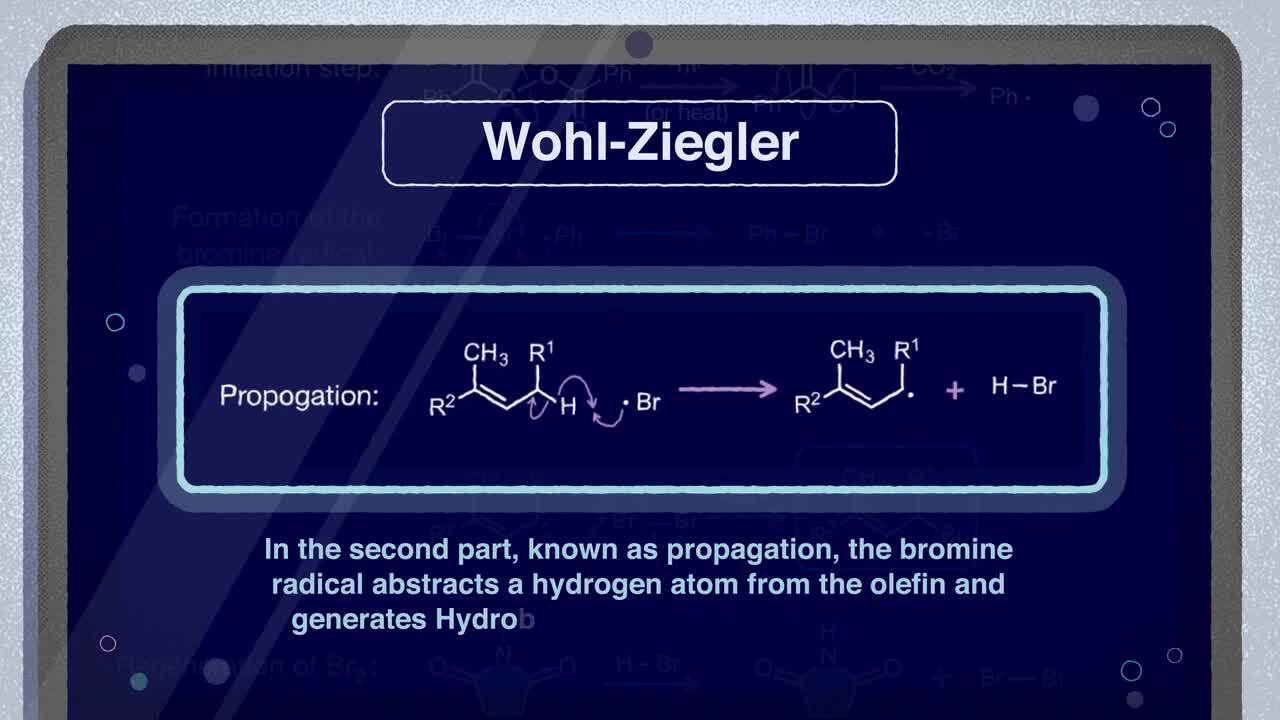
Wohl-Ziegler Bromination Mechanism
Watch our video to learn more about the Wohl-Ziegler bromination and its mechanisms.
History of the Wohl-Ziegler Bromination
In 1919, German chemist Alfred Wohl investigated the reaction between 2,3-dimethyl-2-butene and N-bromoacetamide in diethyl ether, and discovered that the double bond of the substrate remained unchanged and one of the methyl groups was replaced by bromine. This discovery was interesting, as the reaction was previously thought to require the use of bromine at high temperatures to react with the alkenes. In 1942, Karl Ziegler carried out a comprehensive study of the utility of N-bromosuccinimide (NBS) in the allylic bromination of olefins and demonstrated the synthetic value of the process. Subsequently, the addition of bromine at the allylic position of olefins or at the benzylic position of alkylated aromatic or heteroaromatic compounds became known as the Wohl-Ziegler bromination.
The Wohl-Ziegler bromination has been utilized in several total synthesis campaigns, including James M. Cook’s preparation of (-)-tryprostatin A, a natural product isolated from a marine fungus that inhibits cell cycle progression.
Other Free Radical Reactions
Other Free Radical Reactions include:
-
Hunsdiecker Reaction -
Keck Radical Allylation -
Meerwein Arylation -
Sandmeyer Reaction
For other types of reactions, visit our Named Reactions page.
Acroseal Packaging
Chemical reactions often involve the use of air- and moisture-sensitive solvents, and pyrophoric or hazardous reagents. Our AcroSeal packaging is a packaging solution designed to enable safe handling of these types of materials which are used in a variety of research and development applications, including NMR analysis and studies in drug discovery, agrochemicals, flavors and fragrances, and more.
Watch our video for more information.