Search Thermo Fisher Scientific
Thermo Scientific Chemicals
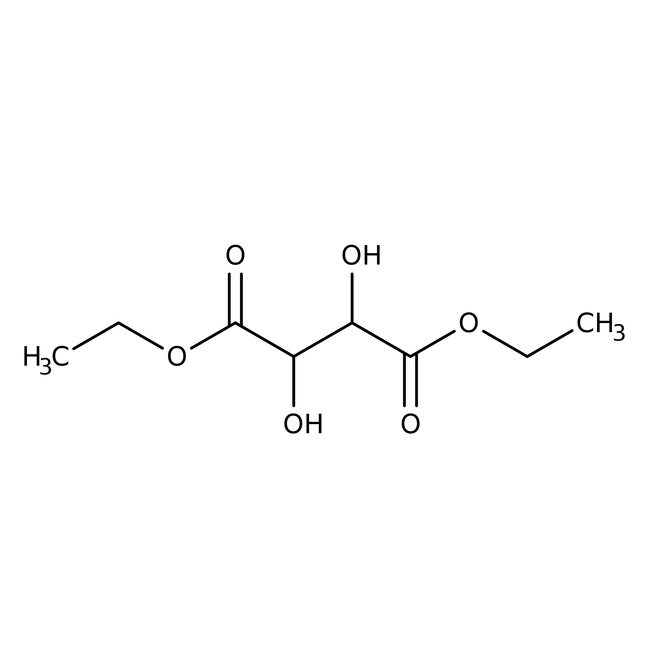
(+)-Diethyl L-tartrate, 98%, Thermo Scientific ChemicalsCatalog number A10641.22
also known as A10641-22
Price (USD)/ Each
42.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
49.80 Save 7.15 (14%)
-
Quantity:
100 g
Price (USD)/ Each
42.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
49.80 Save 7.15 (14%)
(+)-Diethyl L-tartrate, 98%, Thermo Scientific Chemicals
Catalog numberA10641.22
Price (USD)/ Each
42.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
49.80 Save 7.15 (14%)
-
Chemical Identifiers
CAS87-91-2
IUPAC Name1,4-diethyl 2,3-dihydroxybutanedioate
Molecular FormulaC8H14O6
InChI KeyYSAVZVORKRDODB-UHFFFAOYNA-N
SMILESCCOC(=O)C(O)C(O)C(=O)OCC
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless
FormLiquid
Identification (FTIR)Conforms
Refractive Index1.4445-1.4485 @ 20?C
Assay (GC)≥98.5%
View more
(+)-Diethyl L-tartrate is used as a chiral auxiliary in the Sharpless enantioselective epoxidation of allylic alcohols, for Sharpless-type enantioselective oxidation of sulfides to sulfoxides and as a chiral auxiliary in the enantioselective construction of cyclopropanes from allylic alcohols by an asymmetric Simmons-Smith reaction. It is also used as a chiral reagent in a host of chemical reactions, such as the synthesis of isoquinoline alkaloids and arundic acid, which has been used in acute ischemic stroke therapy.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
(+)-Diethyl L-tartrate is used as a chiral auxiliary in the Sharpless enantioselective epoxidation of allylic alcohols, for Sharpless-type enantioselective oxidation of sulfides to sulfoxides and as a chiral auxiliary in the enantioselective construction of cyclopropanes from allylic alcohols by an asymmetric Simmons-Smith reaction. It is also used as a chiral reagent in a host of chemical reactions, such as the synthesis of isoquinoline alkaloids and arundic acid, which has been used in acute ischemic stroke therapy.
Solubility
Fully miscible in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents.
(+)-Diethyl L-tartrate is used as a chiral auxiliary in the Sharpless enantioselective epoxidation of allylic alcohols, for Sharpless-type enantioselective oxidation of sulfides to sulfoxides and as a chiral auxiliary in the enantioselective construction of cyclopropanes from allylic alcohols by an asymmetric Simmons-Smith reaction. It is also used as a chiral reagent in a host of chemical reactions, such as the synthesis of isoquinoline alkaloids and arundic acid, which has been used in acute ischemic stroke therapy.
Solubility
Fully miscible in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents.
RUO – Research Use Only
Peter Somfai. An enantioselective total synthesis of (+)-altholactone from diethyl L-tartrate. Tetrahedron. 1994, 50, (38), 11315-11320.
Woo-Wha Lee.; Sukbok Chang. A new and concise synthetic route to an enantiopure (+)-conduritol-E derivative from diethyl l-tartrate. Tetrahedron: Asymmetry. 1999, 10 (23), 4473-4475.
For use as a chiral auxiliary in the Sharpless enantioselective epoxidation of allylic alcohols, see tert-Butyl hydroperoxide, A13926. For Sharpless-type enantioselective oxidation of sulfides to sulfoxides, see: Org. Synth. Coll., 8, 464 (1993).
For use as a chiral auxiliary in the enantioselective construction of cyclopropanes from allylic alcohols by an asymmetric Simmons-Smith reaction, see: Chem. Lett., 61 (1992). For a review of the use of tartrate esters as chiral auxiliaries, e.g. in the Simmons-Smith reaction and in 1,3-dipolar cycloaddition reactions, see: Synlett, 1075 (2003).
Woo-Wha Lee.; Sukbok Chang. A new and concise synthetic route to an enantiopure (+)-conduritol-E derivative from diethyl l-tartrate. Tetrahedron: Asymmetry. 1999, 10 (23), 4473-4475.
For use as a chiral auxiliary in the Sharpless enantioselective epoxidation of allylic alcohols, see tert-Butyl hydroperoxide, A13926. For Sharpless-type enantioselective oxidation of sulfides to sulfoxides, see: Org. Synth. Coll., 8, 464 (1993).
For use as a chiral auxiliary in the enantioselective construction of cyclopropanes from allylic alcohols by an asymmetric Simmons-Smith reaction, see: Chem. Lett., 61 (1992). For a review of the use of tartrate esters as chiral auxiliaries, e.g. in the Simmons-Smith reaction and in 1,3-dipolar cycloaddition reactions, see: Synlett, 1075 (2003).