Search Thermo Fisher Scientific
Thermo Scientific Chemicals
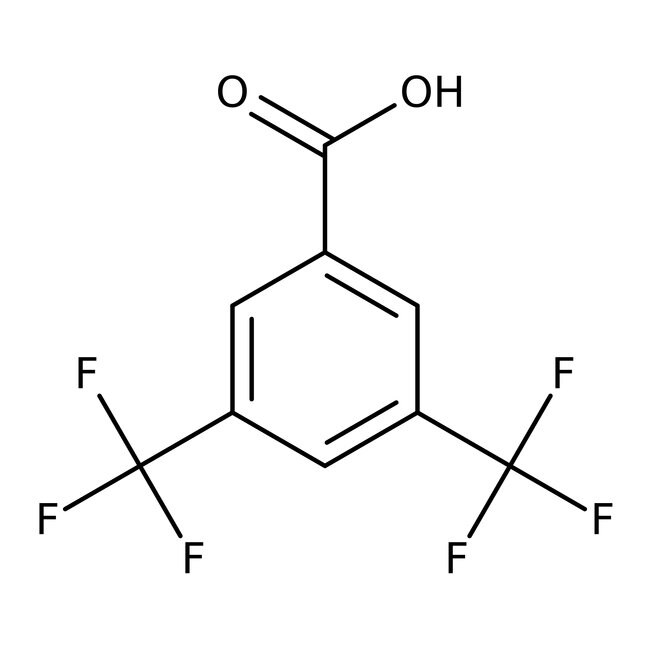
3,5-Bis(trifluoromethyl)benzoic acid, 98%, Thermo Scientific ChemicalsCatalog number A11325.06
also known as A11325-06
Price (USD)/ Each
58.10
-
Quantity:
5 g
Price (USD)/ Each
58.10
3,5-Bis(trifluoromethyl)benzoic acid, 98%, Thermo Scientific Chemicals
Catalog numberA11325.06
Price (USD)/ Each
58.10
-
Chemical Identifiers
CAS725-89-3
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to pale cream
Melting Point (clear melt)140.0-147.0?C
Assay (Aqueous acid-base Titration)≥97.5 to ≤102.5%
FormCrystals or powder or crystalline powder
Assay (Silylated GC)≥97.5%
In vitro metabolism studies on a series of 3,5-bis(trifluoromethyl)benzyl ethers have identified 3,5-bis(trifluoromethyl)benzoic acid as a significant metabolite possibly arising via oxidation of the benzylic position. The reactivity of the hydroxy- propyl units was demonstrated by the esterification of por- phyrazine derivatives with 3,5-bis(trifluoromethyl)benzoic acid in the presence of dicyclohexylcarbodiimide and toluene- p-sulfonic acid.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
In vitro metabolism studies on a series of 3,5-bis(trifluoromethyl)benzyl ethers have identified 3,5-bis(trifluoromethyl)benzoic acid as a significant metabolite possibly arising via oxidation of the benzylic position. The reactivity of the hydroxy- propyl units was demonstrated by the esterification of por- phyrazine derivatives with 3,5-bis(trifluoromethyl)benzoic acid in the presence of dicyclohexylcarbodiimide and toluene- p-sulfonic acid.
Solubility
Slightly soluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
In vitro metabolism studies on a series of 3,5-bis(trifluoromethyl)benzyl ethers have identified 3,5-bis(trifluoromethyl)benzoic acid as a significant metabolite possibly arising via oxidation of the benzylic position. The reactivity of the hydroxy- propyl units was demonstrated by the esterification of por- phyrazine derivatives with 3,5-bis(trifluoromethyl)benzoic acid in the presence of dicyclohexylcarbodiimide and toluene- p-sulfonic acid.
Solubility
Slightly soluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
RUO – Research Use Only
General References:
- Swain CJ, et al. 3-Benzyloxy-2-phenylpiperidine NK1 antagonists: the influence of alpha methyl substitution.. Bioorg. Med. Chem.Lett. 1997, 7(23), 2959-62.
- Tuğba Şimşek,; Ergün Gonca. Enrichment of solubility by esterification: Metal-free and metallo-porphyrazines with polyfluorinated units. Transition Metal Chemistry. 2013, 38(1), 37-43.