Search Thermo Fisher Scientific
Thermo Scientific Chemicals
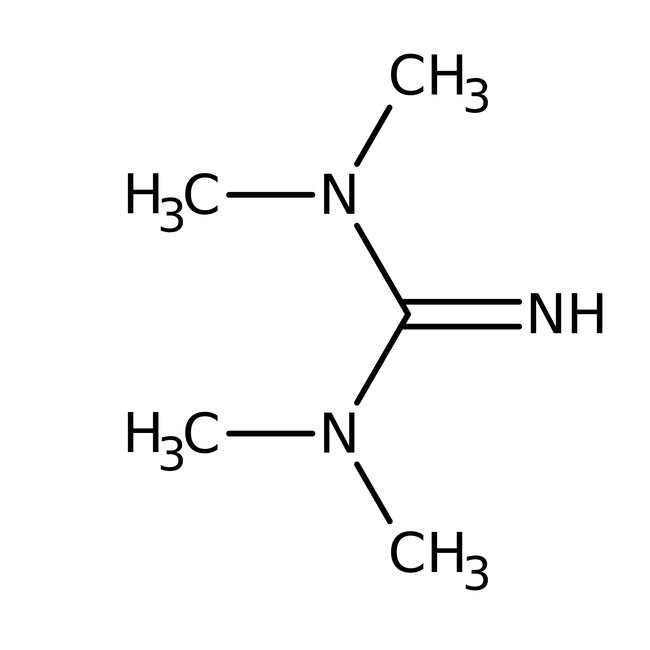
1,1,3,3-Tetramethylguanidine, 99%, Thermo Scientific ChemicalsCatalog number A12314.AE
also known as A12314-AE
Price (USD)/ Each
58.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
69.20 Save 10.55 (15%)
-
Quantity:
100 mL
Price (USD)/ Each
58.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
69.20 Save 10.55 (15%)
1,1,3,3-Tetramethylguanidine, 99%, Thermo Scientific Chemicals
Catalog numberA12314.AE
Price (USD)/ Each
58.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
69.20 Save 10.55 (15%)
-
Chemical Identifiers
CAS80-70-6
IUPAC NameN,N,N',N'-tetramethylguanidine
Molecular FormulaC5H13N3
InChI KeyKYVBNYUBXIEUFW-UHFFFAOYSA-N
SMILESCN(C)C(=N)N(C)C
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless to yellow
Assay (GC)≥98.5% (UK sourced material)
Assay from Supplier's CofA≥98.5% (US sourced material, GC)
Refractive Index1.4665-1.4695 @ 20?C (UK sourced material)
FormLiquid
View more
1,1,3,3-Tetramethylguanidine is employed as a polyurethane foam catalyst, as an accelerator for the syntheses of polysulfured rubber. It is also used as a strong base in the photochemical (couplers) and in the pharmaceutical (steroids) industries. It is essential for the preparation of alkyl nitriles from alkyl halides and 3'-alkylthymidines from 3'-nitrothymidines. It serves as an efficient and selective catalyst for the benzoylation of alcohols. It replaces the expensive bases 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) and 1,5-Diazabicyclo[4.3.0]non-5-ene (DBN) in organic synthesis.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1,1,3,3-Tetramethylguanidine is employed as a polyurethane foam catalyst, as an accelerator for the syntheses of polysulfured rubber. It is also used as a strong base in the photochemical (couplers) and in the pharmaceutical (steroids) industries. It is essential for the preparation of alkyl nitriles from alkyl halides and 3′-alkylthymidines from 3′-nitrothymidines. It serves as an efficient and selective catalyst for the benzoylation of alcohols. It replaces the expensive bases 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) and 1,5-Diazabicyclo[4.3.0]non-5-ene (DBN) in organic synthesis.
Solubility
Miscible with water.
Notes
Air sensitive. Incompatible with strong oxidizing agents, mineral and organic acids, carbon dioxide. Keep away from sources of ignition.
1,1,3,3-Tetramethylguanidine is employed as a polyurethane foam catalyst, as an accelerator for the syntheses of polysulfured rubber. It is also used as a strong base in the photochemical (couplers) and in the pharmaceutical (steroids) industries. It is essential for the preparation of alkyl nitriles from alkyl halides and 3′-alkylthymidines from 3′-nitrothymidines. It serves as an efficient and selective catalyst for the benzoylation of alcohols. It replaces the expensive bases 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) and 1,5-Diazabicyclo[4.3.0]non-5-ene (DBN) in organic synthesis.
Solubility
Miscible with water.
Notes
Air sensitive. Incompatible with strong oxidizing agents, mineral and organic acids, carbon dioxide. Keep away from sources of ignition.
RUO – Research Use Only
Strong organic base useful for the formation of organic-soluble salts, and as a basic catalyst, e.g. in Michael addition reactions: J. Org. Chem., 27, 3175 (1962); Tetrahedron Lett., 569 (1973). Preferred catalyst in the Michael addition of aliphatic nitro compounds to unsaturated esters: Synthesis, 44 (1972); 953 (1989); Tetrahedron Lett., 30, 993 (1989).
Useful base for formation of TBDMS ethers of alcohols with tert-Butyldimethylchlorosilane, A13064: J. Org. Chem., 49, 4657 (1984).
For use in the Baylis-Hillman reaction, see: J. Chem. Soc., Perkin 1, 2831 (2001).
Inexpensive and efficient ligand for the palladium-catalyzed Heck reaction: Synlett, 1885 (2005).
Other uses include: Acylation of amino acids by methyl trifluoroacetate: Synthesis, 399 (1976). Formation of Boc-amino acids with tert-butyl phenyl carbonate: Org. Synth. Coll., 6, 203 (1988). Cleavage of peptides from resins: Tetrahedron, 40, 4237 (1984). Selective cleavage of a primary benzyloxycarbonyl (Cbz, Z) group in the presence of either Boc or secondary Cbz: J. Chem. Soc., Perkin 1, 1905 (1988). See also 4-Chloromethylpyridine hydrochloride, A12859 and Appendix 6.
Liang, S.; Zhou, Y.; Liu, H.; Jiang, T.; Han, B. Immobilized 1,1,3,3-Tetramethylguanidine Ionic Liquids as the Catalyst for Synthesizing Propylene Glycol Methyl Ether. Catal. Lett. 2010, 140 (1-2), 49-54.
Huczynski, A.; Janczak, J.; Stefanska, J.; Rutkowski, J.; Brzezinski, B. X-ray, spectroscopic and antibacterial activity studies of the 1:1 complex of lasalocid acid with 1,1,3,3-tetramethylguanidine. J. Mol. Struct. 2010, 977 (1-3), 51-55.
Useful base for formation of TBDMS ethers of alcohols with tert-Butyldimethylchlorosilane, A13064: J. Org. Chem., 49, 4657 (1984).
For use in the Baylis-Hillman reaction, see: J. Chem. Soc., Perkin 1, 2831 (2001).
Inexpensive and efficient ligand for the palladium-catalyzed Heck reaction: Synlett, 1885 (2005).
Other uses include: Acylation of amino acids by methyl trifluoroacetate: Synthesis, 399 (1976). Formation of Boc-amino acids with tert-butyl phenyl carbonate: Org. Synth. Coll., 6, 203 (1988). Cleavage of peptides from resins: Tetrahedron, 40, 4237 (1984). Selective cleavage of a primary benzyloxycarbonyl (Cbz, Z) group in the presence of either Boc or secondary Cbz: J. Chem. Soc., Perkin 1, 1905 (1988). See also 4-Chloromethylpyridine hydrochloride, A12859 and Appendix 6.
Liang, S.; Zhou, Y.; Liu, H.; Jiang, T.; Han, B. Immobilized 1,1,3,3-Tetramethylguanidine Ionic Liquids as the Catalyst for Synthesizing Propylene Glycol Methyl Ether. Catal. Lett. 2010, 140 (1-2), 49-54.
Huczynski, A.; Janczak, J.; Stefanska, J.; Rutkowski, J.; Brzezinski, B. X-ray, spectroscopic and antibacterial activity studies of the 1:1 complex of lasalocid acid with 1,1,3,3-tetramethylguanidine. J. Mol. Struct. 2010, 977 (1-3), 51-55.