Search Thermo Fisher Scientific
Thermo Scientific Chemicals
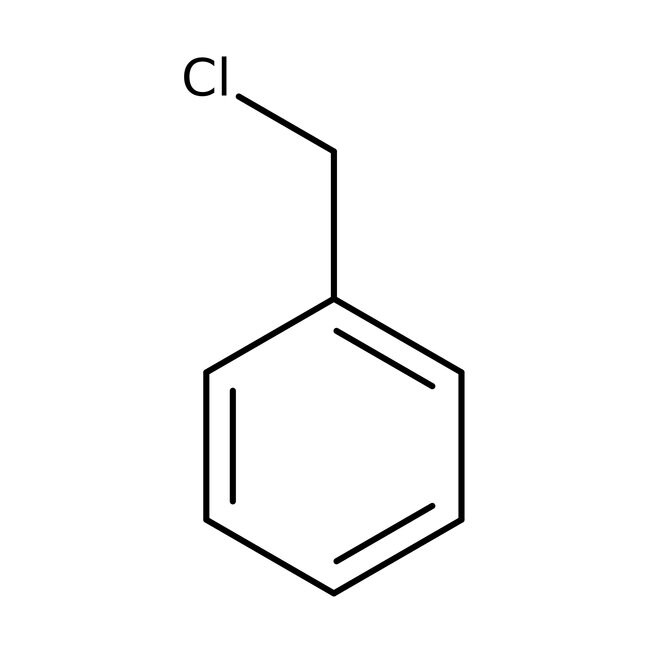
Benzyl chloride, 99%, stab., Thermo Scientific ChemicalsCatalog number A12481.0E
also known as A12481-0E
Price (USD)/ Each
106.00
-
Quantity:
2500 g
Price (USD)/ Each
106.00
Benzyl chloride, 99%, stab., Thermo Scientific Chemicals
Catalog numberA12481.0E
Price (USD)/ Each
106.00
-
Chemical Identifiers
CAS100-44-7
IUPAC Name(chloromethyl)benzene
Molecular FormulaC7H7Cl
InChI KeyKCXMKQUNVWSEMD-UHFFFAOYSA-N
SMILESClCC1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless to pale yellow
FormLiquid
Assay (GC)≥98.5% (non-U.S. specifiaction)
Assay from Suppliers CofA≥98.0% (GC, U.S specification)
CommentPurchased in the U.S. and in other countries
View more
Benzyl Chloride is used as an intermediate for manufacturing of benzyl alcohol, benzoic acid and benzyl cyanide. It is also used to prepare benzyl esters, which are used as plasticizer, flavorants and perfumes. It acts as an intermediate in the preparation of phenylacetic acid. In organic synthesis, it is used as an alkylating agent and utilized for the introduction of the benzyl protecting group for alcohols. Further, it is involved in the preparation of quaternary ammonium salts by reaction with tertiary amines. In addition to this, it is used to prepare Grignard reagent by reacting with metallic magnesium.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Benzyl Chloride is used as an intermediate for manufacturing of benzyl alcohol, benzoic acid and benzyl cyanide. It is also used to prepare benzyl esters, which are used as plasticizer, flavorants and perfumes. It acts as an intermediate in the preparation of phenylacetic acid. In organic synthesis, it is used as an alkylating agent and utilized for the introduction of the benzyl protecting group for alcohols. Further, it is involved in the preparation of quaternary ammonium salts by reaction with tertiary amines. In addition to this, it is used to prepare Grignard reagent by reacting with metallic magnesium.
Solubility
Miscible with alcohol, ether. Insoluble in waterMiscible with alcohol, ether and chloroform.
Notes
Light and moisture sensitive. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with common metals, lead,oxidizing agents, iron, iron salts, brass and aluminum
Benzyl Chloride is used as an intermediate for manufacturing of benzyl alcohol, benzoic acid and benzyl cyanide. It is also used to prepare benzyl esters, which are used as plasticizer, flavorants and perfumes. It acts as an intermediate in the preparation of phenylacetic acid. In organic synthesis, it is used as an alkylating agent and utilized for the introduction of the benzyl protecting group for alcohols. Further, it is involved in the preparation of quaternary ammonium salts by reaction with tertiary amines. In addition to this, it is used to prepare Grignard reagent by reacting with metallic magnesium.
Solubility
Miscible with alcohol, ether. Insoluble in waterMiscible with alcohol, ether and chloroform.
Notes
Light and moisture sensitive. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with common metals, lead,oxidizing agents, iron, iron salts, brass and aluminum
WARNING: Cancer – www.P65Warnings.ca.gov
RUO – Research Use Only
Benzyllithium and substituted analogues have proved difficult to generate in a pure state owing to competing Wurtz coupling. Conditions have been described for efficient generation of benzyllithiums from the chlorides using Li naphthalenide in a mixed diethyl ether - THF - petroleum ether solvent system at -95o: J. Chem. Soc., Perkin 1, 185 (1995).
Amidocarbonylation with acetamide and a CO/H2 mixture under pressure, in the presence of Co2(CO)8 catalyst, provides a novel route to N-acetyl-DL-phenylalanine: J. Org. Chem., 61, 1842 (1996).
Lázaro, G.; Polo, V.; Fernández-Alvarez, F. J.; García-Orduña, P.; Lahoz, F. J.; Iglesias, M.; Pérez-Torrente, J. J.; Oro, L. A. Catalytic Hydrodechlorination of Benzyl Chloride Promoted by Rh-N-heterocyclic Carbene Catalysts. ChemSusChem 2015, 8 (3), 495-503.
Rodda, A. E.; Ercole, F.; Nisbet, D. R.; Forsythe, J. S.; Meagher, L. Optimization of Aqueous SI-ATRP Grafting of Poly(Oligo(Ethylene Glycol) Methacrylate) Brushes from Benzyl Chloride Macroinitiator Surfaces. Macromol. Biosci. 2015, 15 (6), 799-811.
Amidocarbonylation with acetamide and a CO/H2 mixture under pressure, in the presence of Co2(CO)8 catalyst, provides a novel route to N-acetyl-DL-phenylalanine: J. Org. Chem., 61, 1842 (1996).
Lázaro, G.; Polo, V.; Fernández-Alvarez, F. J.; García-Orduña, P.; Lahoz, F. J.; Iglesias, M.; Pérez-Torrente, J. J.; Oro, L. A. Catalytic Hydrodechlorination of Benzyl Chloride Promoted by Rh-N-heterocyclic Carbene Catalysts. ChemSusChem 2015, 8 (3), 495-503.
Rodda, A. E.; Ercole, F.; Nisbet, D. R.; Forsythe, J. S.; Meagher, L. Optimization of Aqueous SI-ATRP Grafting of Poly(Oligo(Ethylene Glycol) Methacrylate) Brushes from Benzyl Chloride Macroinitiator Surfaces. Macromol. Biosci. 2015, 15 (6), 799-811.