Search Thermo Fisher Scientific
Thermo Scientific Chemicals
2,2-Dimethoxypropane, 98%, Thermo Scientific ChemicalsCatalog number A13810.0F
also known as A13810-0F
Price (USD)/ Each
138.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
163.00 Save 24.35 (15%)
-
Quantity:
2500 mL
Price (USD)/ Each
138.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
163.00 Save 24.35 (15%)
2,2-Dimethoxypropane, 98%, Thermo Scientific Chemicals
Catalog numberA13810.0F
Price (USD)/ Each
138.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
163.00 Save 24.35 (15%)
-
Chemical Identifiers
CAS77-76-9
IUPAC Name2,2-dimethoxypropane
Molecular FormulaC5H12O2
InChI KeyHEWZVZIVELJPQZ-UHFFFAOYSA-N
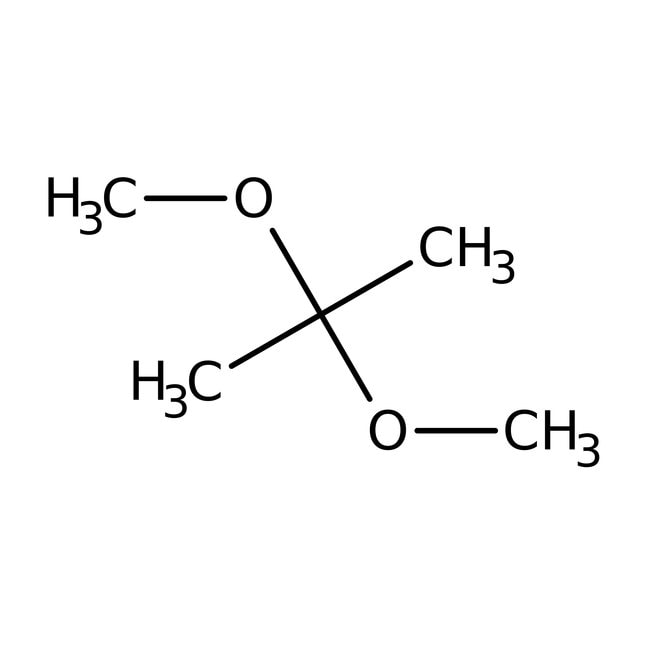
SMILESCOC(C)(C)OC
View more
Specifications Specification Sheet
Specification Sheet
Assay (GC)≥97.5%
Refractive Index1.3755-1.3795 @ 20?C (UK sourced material only)
Appearance (Color)Clear colorless
FormLiquid
Identification (FTIR)Conforms (UK sourced material only)
2,2-Dimethoxypropane acts as a dehydrating agent. It also serves as an intermediate in the synthesis of vitamin E, vitamin A and various carotenoids such as astaxanthin. It is used as a reagent for the preparation of 1,2-diols, acetonides, isopropylidene derivatives of sugars, nucleosides, methyl esters of amino acids and enol ethers.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2,2-Dimethoxypropane acts as a dehydrating agent. It also serves as an intermediate in the synthesis of vitamin E, vitamin A and various carotenoids such as astaxanthin. It is used as a reagent for the preparation of 1,2-diols, acetonides, isopropylidene derivatives of sugars, nucleosides, methyl esters of amino acids and enol ethers.
Solubility
Soluble in benzene, carbon tetrachloride, ethyl ether and n-butane, methanol. Moderately soluble in water.
Notes
Incompatible with oxidizing agents and acids.
2,2-Dimethoxypropane acts as a dehydrating agent. It also serves as an intermediate in the synthesis of vitamin E, vitamin A and various carotenoids such as astaxanthin. It is used as a reagent for the preparation of 1,2-diols, acetonides, isopropylidene derivatives of sugars, nucleosides, methyl esters of amino acids and enol ethers.
Solubility
Soluble in benzene, carbon tetrachloride, ethyl ether and n-butane, methanol. Moderately soluble in water.
Notes
Incompatible with oxidizing agents and acids.
RUO – Research Use Only
Methoxylating agent for preparation of acetals by acid-catalyzed exchange: J. Org. Chem., 25, 521 (1960). Also used as a water-scavenger in the preparation of acetals from higher-boiling alcohols and ketones: Org. Synth. Coll., 5, 292 (1973).
Widely used for the isopropylidenation of diols; see, e.g.: Org. Synth. Coll., 9, 450, 717 (1998). Isopropylidenation of acid-sensitive carbohydrates in the presence of 2,3-Dichloro-5,6-dicyanobenzoquinone, A11879: Acta Chem. Scand., 47, 843 (1993); preparation of acyclic mixed acetals of carbohydrates: Acta Chem. Scand., 48, 80 (1994). For use in the cyclization of N-Boc serine methyl ester to the oxazolidine, see: Org. Synth. Coll., 9, 300 (1998).
Methyl ester hydrochlorides of amino acids can be conveniently prepared with excess reagent in combination with conc. HCl. Water is removed in situ, thus displacing the equilibrium in favor of the ester: J. Org. Chem., 28, 3898 (1963). Methyl esters of non-aromatic acids can also be prepared with the reagent in MeOH with a catalytic amount of HCl or TMS chloride: Tetrahedron Lett., 38, 2685 (1997).
Urosa, A.; Marcos, I. S.; Díez, D.; Padrón, J. M.; Pilar Basabe, P. Synthesis of Luffarin L and 16-epi-Luffarin L Using a Temporary Silicon-Tethered Ring-Closing Metathesis Reaction. J. Org. Chem. 2015, 80 (12), 6447-6455.
Widely used for the isopropylidenation of diols; see, e.g.: Org. Synth. Coll., 9, 450, 717 (1998). Isopropylidenation of acid-sensitive carbohydrates in the presence of 2,3-Dichloro-5,6-dicyanobenzoquinone, A11879: Acta Chem. Scand., 47, 843 (1993); preparation of acyclic mixed acetals of carbohydrates: Acta Chem. Scand., 48, 80 (1994). For use in the cyclization of N-Boc serine methyl ester to the oxazolidine, see: Org. Synth. Coll., 9, 300 (1998).
Methyl ester hydrochlorides of amino acids can be conveniently prepared with excess reagent in combination with conc. HCl. Water is removed in situ, thus displacing the equilibrium in favor of the ester: J. Org. Chem., 28, 3898 (1963). Methyl esters of non-aromatic acids can also be prepared with the reagent in MeOH with a catalytic amount of HCl or TMS chloride: Tetrahedron Lett., 38, 2685 (1997).
Urosa, A.; Marcos, I. S.; Díez, D.; Padrón, J. M.; Pilar Basabe, P. Synthesis of Luffarin L and 16-epi-Luffarin L Using a Temporary Silicon-Tethered Ring-Closing Metathesis Reaction. J. Org. Chem. 2015, 80 (12), 6447-6455.