Search Thermo Fisher Scientific
Thermo Scientific Chemicals
Salicylaldehyde, 99%, Thermo Scientific ChemicalsCatalog number A13833.36
also known as A13833-36
Price (USD)/ Each
69.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
81.60 Save 11.95 (15%)
-
Quantity:
500 g
Price (USD)/ Each
69.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
81.60 Save 11.95 (15%)
Salicylaldehyde, 99%, Thermo Scientific Chemicals
Catalog numberA13833.36
Price (USD)/ Each
69.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
81.60 Save 11.95 (15%)
-
Chemical Identifiers
CAS90-02-8
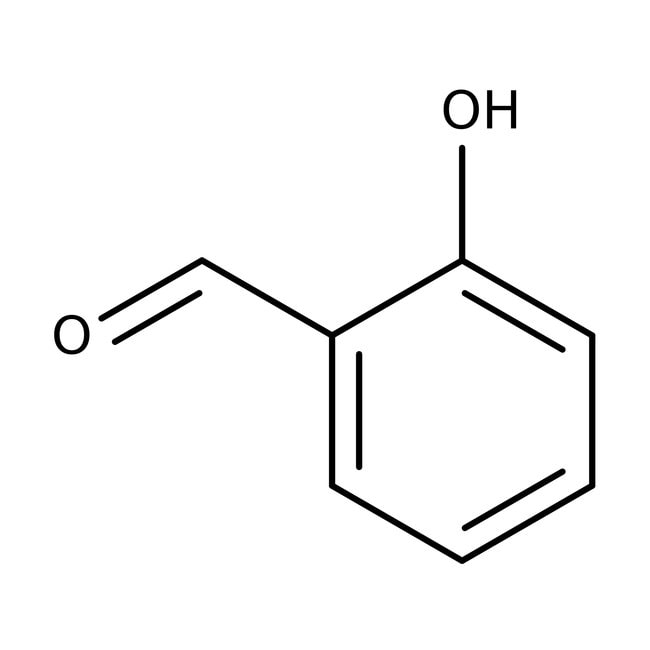
IUPAC Name2-hydroxybenzaldehyde
Molecular FormulaC7H6O2
InChI KeySMQUZDBALVYZAC-UHFFFAOYSA-N
SMILESOC1=CC=CC=C1C=O
View more
Specifications Specification Sheet
Specification Sheet
FormLiquid
Refractive Index1.5715-1.5745 @ 20?C
Appearance (Color)Clear, colourless to pale yellow
Assay (GC)≥98.5%
Impurity contentPhenol: ≤1.0%
View more
Precursor to a variety of chelating agents.Salicylaldehyde is used as a precursor to prepare catechol, benzofuran, salicylaldehydimine and 3-carbethoxycoumarin. It is used for colorimetric assay of isoleucine as well as utilized for the determination of primary amine by fluorescence method. It is a key starting material to a various chelating agents. It is used as flavor and fragrance components. Its derivative salicylaldehyde thiosemicarbazone is used for the simultaneous determination of molybdenum and iron by spectrophotometric method.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Precursor to a variety of chelating agents.Salicylaldehyde is used as a precursor to prepare catechol, benzofuran, salicylaldehydimine and 3-carbethoxycoumarin. It is used for colorimetric assay of isoleucine as well as utilized for the determination of primary amine by fluorescence method. It is a key starting material to a various chelating agents. It is used as flavor and fragrance components. Its derivative salicylaldehyde thiosemicarbazone is used for the simultaneous determination of molybdenum and iron by spectrophotometric method.
Solubility
Miscible with alcohol, ether, acetone, benzene and most organic solvents. Slightly miscible with water.
Notes
Incompatible with acids, strong reducing agents, strong oxidizing agents, strong bases and halogens.
Precursor to a variety of chelating agents.Salicylaldehyde is used as a precursor to prepare catechol, benzofuran, salicylaldehydimine and 3-carbethoxycoumarin. It is used for colorimetric assay of isoleucine as well as utilized for the determination of primary amine by fluorescence method. It is a key starting material to a various chelating agents. It is used as flavor and fragrance components. Its derivative salicylaldehyde thiosemicarbazone is used for the simultaneous determination of molybdenum and iron by spectrophotometric method.
Solubility
Miscible with alcohol, ether, acetone, benzene and most organic solvents. Slightly miscible with water.
Notes
Incompatible with acids, strong reducing agents, strong oxidizing agents, strong bases and halogens.
RUO – Research Use Only
A convenient method for the racemization of amino acids consists in heating in acetic acid with 0.05 equivalents of an aldehyde, such as salicylaldehyde: J. Org. Chem., 48, 843 (1983).
For a review of the use of circular dichroism studies of salicylidene derivatives of chiral amines, to establish their absolute configurations using the “salicylidene chirality rule”, see: Chem. Rev., 83, 359 (1983).
Salem, N. M.; Rashad, A. R.; El Sayed, L.; Foro, S.; Haase, W.; Iskander, M. F. Synthesis, characterization, molecular structure and supramolecular architectures of some copper(II) complexes derived from salicylaldehyde semicarbazone. Inorg. Chim. Acta 2015, 432, 231-242.
Li, K.; Xiang, Y.; Wang, X.; Li, J.; Hu, R.; Tong, A.; Tang, B. Z. Reversible photochromic system based on rhodamine B salicylaldehyde hydrazone metal complex. J. Am. Chem. Soc. 2014, 136 (4), 1643-1649.
For a review of the use of circular dichroism studies of salicylidene derivatives of chiral amines, to establish their absolute configurations using the “salicylidene chirality rule”, see: Chem. Rev., 83, 359 (1983).
Salem, N. M.; Rashad, A. R.; El Sayed, L.; Foro, S.; Haase, W.; Iskander, M. F. Synthesis, characterization, molecular structure and supramolecular architectures of some copper(II) complexes derived from salicylaldehyde semicarbazone. Inorg. Chim. Acta 2015, 432, 231-242.
Li, K.; Xiang, Y.; Wang, X.; Li, J.; Hu, R.; Tong, A.; Tang, B. Z. Reversible photochromic system based on rhodamine B salicylaldehyde hydrazone metal complex. J. Am. Chem. Soc. 2014, 136 (4), 1643-1649.