Search Thermo Fisher Scientific
Thermo Scientific Chemicals
3-Sulfolene, 98%, Thermo Scientific ChemicalsCatalog number A13887.0I
also known as A13887-0I
Price (USD)/ Each
327.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
385.00 Save 57.35 (15%)
-
Quantity:
5000 g
Price (USD)/ Each
327.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
385.00 Save 57.35 (15%)
3-Sulfolene, 98%, Thermo Scientific Chemicals
Catalog numberA13887.0I
Price (USD)/ Each
327.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
385.00 Save 57.35 (15%)
-
Chemical Identifiers
CAS77-79-2
IUPAC Name2,5-dihydro-1λ⁶-thiophene-1,1-dione
Molecular FormulaC4H6O2S
InChI KeyMBDNRNMVTZADMQ-UHFFFAOYSA-N
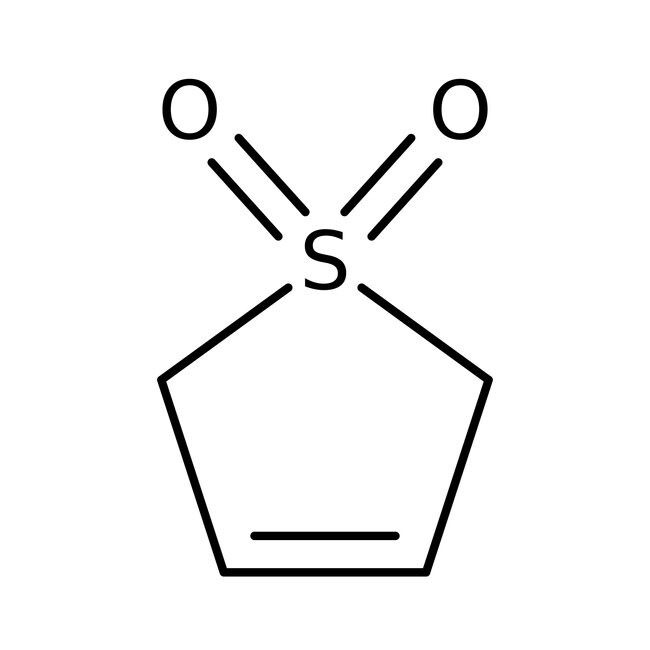
SMILESO=S1(=O)CC=CC1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to cream
FormCrystals or powder or crystalline powder or lumps or chunks
Melting Point (clear melt)62.0-68.0?C
Assay (GC)≥97.5%
It is a source of cisoid butadiene for Diels-Alder reactions. This chemical is also a convenient source of sulfur dioxide. It is a solvent used in the petrochemical industry for the extraction of aromatics from hydrocarbon streams.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
It is a source of cisoid butadiene for Diels-Alder reactions. This chemical is also a convenient source of sulfur dioxide. It is a solvent used in the petrochemical industry for the extraction of aromatics from hydrocarbon streams.
Solubility
Soluble in water, ethanol, benzene, ether.
Notes
Store in a cool, dry, well-ventilated area. Store away from oxidizing agent.
It is a source of cisoid butadiene for Diels-Alder reactions. This chemical is also a convenient source of sulfur dioxide. It is a solvent used in the petrochemical industry for the extraction of aromatics from hydrocarbon streams.
Solubility
Soluble in water, ethanol, benzene, ether.
Notes
Store in a cool, dry, well-ventilated area. Store away from oxidizing agent.
RUO – Research Use Only
Thomas E. SampleJr.; and Lewis F. Hatch. 3-Sulfolene: A butadiene source for a Diels-Alder synthesis: An undergraduate laboratory experiment. J. Chem. Educ. 1968, 45 (1), 55.
Hans-Joachim Lehmler. Synthesis of environmentally relevant fluorinated surfactants—a review. Chemosphere. 2005, 58 (11), 1471-1496.
Convenient crystalline source of 1,3-butadiene, which is generated on heating to about 110oC: Rec. Trav. Chim., 61, 785 (1942). For an example of its use in the Diels-Alder reaction, see: Org. Synth. Coll., 6, 454 (1988).
Also behaves as a dienophile in with reactive dienes, for example 1,3-Diphenylisobenzofuran, L00101: J. Org. Chem., 34, 538 (1969).
In the presence of Pd(OAc)2, couples with aryldiazonium fluoroborates to give 3-aryl-4-sulfolenes which are readily isomerized with triethylamine to the 3-aryl-3-sulfolenes, providing a source of the corresponding 2-arylbutadienes: Synth. Commun., 26, 231 (1996).
Lithiation occurs at the 2-position; subsequent alkylation and thermal extrusion of SO2 gave a polyene intermediate in a synthesis of the taxane ring system: J. Org. Chem., 62, 2957 (1997).
Hans-Joachim Lehmler. Synthesis of environmentally relevant fluorinated surfactants—a review. Chemosphere. 2005, 58 (11), 1471-1496.
Convenient crystalline source of 1,3-butadiene, which is generated on heating to about 110oC: Rec. Trav. Chim., 61, 785 (1942). For an example of its use in the Diels-Alder reaction, see: Org. Synth. Coll., 6, 454 (1988).
Also behaves as a dienophile in with reactive dienes, for example 1,3-Diphenylisobenzofuran, L00101: J. Org. Chem., 34, 538 (1969).
In the presence of Pd(OAc)2, couples with aryldiazonium fluoroborates to give 3-aryl-4-sulfolenes which are readily isomerized with triethylamine to the 3-aryl-3-sulfolenes, providing a source of the corresponding 2-arylbutadienes: Synth. Commun., 26, 231 (1996).
Lithiation occurs at the 2-position; subsequent alkylation and thermal extrusion of SO2 gave a polyene intermediate in a synthesis of the taxane ring system: J. Org. Chem., 62, 2957 (1997).