Search Thermo Fisher Scientific
Thermo Scientific Chemicals
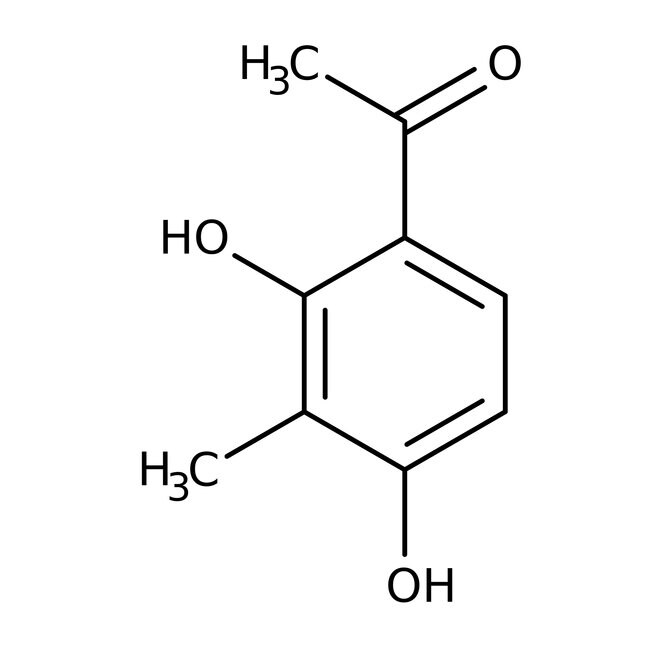
2',4'-Dihydroxy-3'-methylacetophenone, 98%, Thermo Scientific Chemicals
Catalog number B20272.06
also known as B20272-06
Price (USD)/ Each
64.80
-
Quantity:
5 g
Price (USD)/ Each
64.80
2',4'-Dihydroxy-3'-methylacetophenone, 98%, Thermo Scientific Chemicals
Catalog numberB20272.06
Price (USD)/ Each
64.80
-
Chemical Identifiers
CAS10139-84-1
IUPAC Name1-(2,4-dihydroxy-3-methylphenyl)ethan-1-one
Molecular FormulaC9H10O3
InChI KeyKMTLZBUHQPQFAV-UHFFFAOYSA-N
SMILESCC(=O)C1=CC=C(O)C(C)=C1O
View more
Specifications Specification Sheet
Specification Sheet
FormPowder
Appearance (Color)Dark cream to pale brown
Assay (GC)≥97.5%
chemo selective benzylation of 2,4-dihydroxy-3-methylacetophenone gave the 4-benzyloxy derivative. 4- Ethyl-6 methyl resorcinol was prepared by Clemmensen reduction of 4-dihydroxy-3- methylacetophenone.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
chemo selective benzylation of 2,4-dihydroxy-3-methylacetophenone gave the 4-benzyloxy derivative. 4- Ethyl-6 methyl resorcinol was prepared by Clemmensen reduction of 4-dihydroxy-3- methylacetophenone.
Notes
Store in cool, dry place in a well sealed container. Store away from oxidizing agents.
chemo selective benzylation of 2,4-dihydroxy-3-methylacetophenone gave the 4-benzyloxy derivative. 4- Ethyl-6 methyl resorcinol was prepared by Clemmensen reduction of 4-dihydroxy-3- methylacetophenone.
Notes
Store in cool, dry place in a well sealed container. Store away from oxidizing agents.
RUO – Research Use Only
General References:
- David W. Gammon; Roger Hunter; Seanette A. Wilson. An efficient synthesis of 7-hydroxy-2,6-dimethylchromeno[3,4-d]oxazol-4-one—a protected fragment of novenamine. Tetrahedron. 2005, 61 (45), 10683-10688.
- J. P. Brown; D. H. Johnson; Alexander Robertson and W. B. Whalley. 448. Some alkylresorcinols. J. Chem. Soc. 1951, 2019-2020.