Search Thermo Fisher Scientific
Thermo Scientific Chemicals
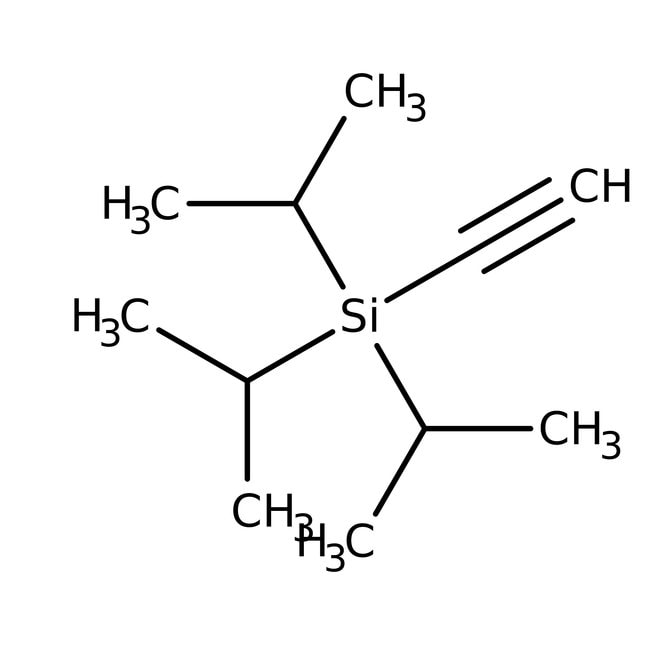
Triisopropylsilylacetylene, 97%, Thermo Scientific ChemicalsCatalog number H53405.06
also known as H53405-06
Price (USD)/ Each
58.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
68.60 Save 9.95 (15%)
-
Quantity:
5 g
Price (USD)/ Each
58.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
68.60 Save 9.95 (15%)
Triisopropylsilylacetylene, 97%, Thermo Scientific Chemicals
Catalog numberH53405.06
Price (USD)/ Each
58.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
68.60 Save 9.95 (15%)
-
Chemical Identifiers
CAS89343-06-6
IUPAC Nameethynyltris(propan-2-yl)silane
Molecular FormulaC11H22Si
InChI KeyKZGWPHUWNWRTEP-UHFFFAOYSA-N
SMILESCC(C)[Si](C#C)(C(C)C)C(C)C
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear, colourless
FormLiquid
Assay (GC)>96.0%
Refractive Index1.4500-1.4550 @ 20?C
Triisopropylsilylacetylene is used as a reagent for the rhodium-catalyzed asymmetric alkylation of alpha, beta-unsaturated carbonyl compounds as well as in the synthesis of beta-alkynylated nitroalkanes. It is utilized in the preparation of bromoethynyl-triisopropyl-silane in the presence of the reagents such as N-bromo succinimide and silver nitrate. It is actively in involved in Sonogashira coupling reaction with 1-bromo-3-iodo-5-tertbutylbenzene.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Triisopropylsilylacetylene is used as a reagent for the rhodium-catalyzed asymmetric alkylation of alpha, beta-unsaturated carbonyl compounds as well as in the synthesis of beta-alkynylated nitroalkanes. It is utilized in the preparation of bromoethynyl-triisopropyl-silane in the presence of the reagents such as N-bromo succinimide and silver nitrate. It is actively in involved in Sonogashira coupling reaction with 1-bromo-3-iodo-5-tertbutylbenzene.
Solubility
Miscible with organic solvents.
Notes
Incompatible with strong acids, strong bases and strong oxidizing agents.
Triisopropylsilylacetylene is used as a reagent for the rhodium-catalyzed asymmetric alkylation of alpha, beta-unsaturated carbonyl compounds as well as in the synthesis of beta-alkynylated nitroalkanes. It is utilized in the preparation of bromoethynyl-triisopropyl-silane in the presence of the reagents such as N-bromo succinimide and silver nitrate. It is actively in involved in Sonogashira coupling reaction with 1-bromo-3-iodo-5-tertbutylbenzene.
Solubility
Miscible with organic solvents.
Notes
Incompatible with strong acids, strong bases and strong oxidizing agents.
RUO – Research Use Only
Sawano, T.; Ashouri, A.; Nishimura, T.; Hayashi, T. Cobalt-Catalyzed Asymmetric 1, 6-Addition of (Triisopropylsilyl)-acetylene to alfa, beta, gamma, delta-Unsaturated Carbonyl Compounds. J. Am. Chem. Soc. 2012, 134 (46), 18936-18939.
Li, Y.; Liu, L.; Kong, D.; Wang, D.; Feng, W.; Yue, T.; Li, C. Palladium-Catalyzed Alkynylation of Morita-Baylis-Hillman Carbonates with (Triisopropylsilyl)acetylene on Water. J. Org. Chem. 2015, 80 (12), 6283-6290.
Li, Y.; Liu, L.; Kong, D.; Wang, D.; Feng, W.; Yue, T.; Li, C. Palladium-Catalyzed Alkynylation of Morita-Baylis-Hillman Carbonates with (Triisopropylsilyl)acetylene on Water. J. Org. Chem. 2015, 80 (12), 6283-6290.