Search Thermo Fisher Scientific
Thermo Scientific Chemicals
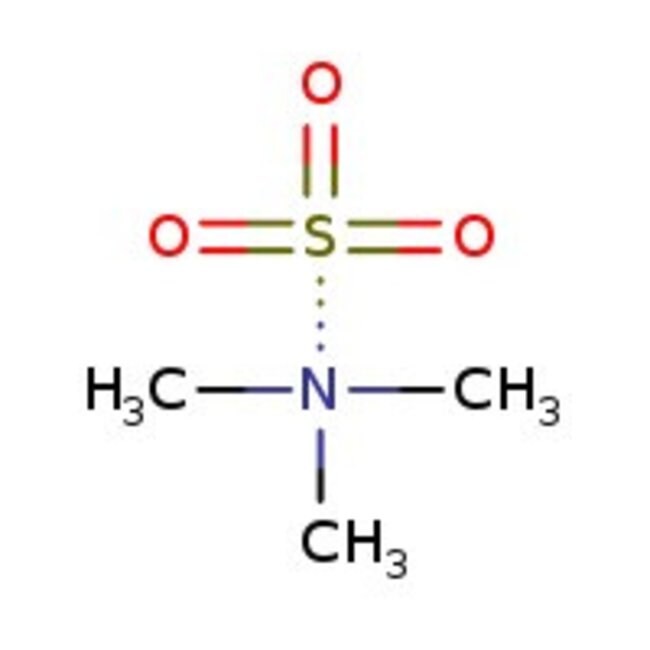
Sulfur trioxide-trimethylamine complex, 95%, Thermo Scientific ChemicalsCatalog number L00302.22
also known as L00302-22
Price (USD)/ Each
106.00
-
Quantity:
100 g
Price (USD)/ Each
106.00
Sulfur trioxide-trimethylamine complex, 95%, Thermo Scientific Chemicals
Catalog numberL00302.22
Price (USD)/ Each
106.00
-
Chemical Identifiers
CAS3162-58-1
IUPAC Namesulfonylideneoxidane; trimethylamine
Molecular FormulaC3H9NO3S
InChI KeyDXASQZJWWGZNSF-UHFFFAOYSA-N
SMILESCN(C)C.O=S(=O)=O
View more
Specifications Specification Sheet
Specification Sheet
Assay (special)≥94.0% (Based on Sulphur elemental analysis)
Identification (FTIR)Conforms
FormPowder
Elemental AnalysisCarbon C: 24.34 - 27.44% (theory 25.89%)
Elemental AnalysisSulphur S: 21.66 - 24.42% (theory 23.04%)
View more
Sulfur trioxide-trimethylamine complex acts as a reagent for sulfonation and sulfamation reactions. It is used as a reactant for the synthesis of sulfate-conjugated resveratrol metabolites and chitooligosaccharides. It is used in association with dodecyl thioglycopyranoside, which acts as a surfactant for enantiomeric separation. Further, it acts as a nucleophile used for the preparation of alfa-tosyloxy ketones.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Sulfur trioxide-trimethylamine complex acts as a reagent for sulfonation and sulfamation reactions. It is used as a reactant for the synthesis of sulfate-conjugated resveratrol metabolites and chitooligosaccharides. It is used in association with dodecyl thioglycopyranoside, which acts as a surfactant for enantiomeric separation. Further, it acts as a nucleophile used for the preparation of alfa-tosyloxy ketones.
Solubility
Soluble in hot water and ethanol. Slightly soluble in dimethyl sulfoxide and methanol. Insoluble in water.
Notes
Moisture sensitive. Incompatible with strong oxidizing agents.
Sulfur trioxide-trimethylamine complex acts as a reagent for sulfonation and sulfamation reactions. It is used as a reactant for the synthesis of sulfate-conjugated resveratrol metabolites and chitooligosaccharides. It is used in association with dodecyl thioglycopyranoside, which acts as a surfactant for enantiomeric separation. Further, it acts as a nucleophile used for the preparation of alfa-tosyloxy ketones.
Solubility
Soluble in hot water and ethanol. Slightly soluble in dimethyl sulfoxide and methanol. Insoluble in water.
Notes
Moisture sensitive. Incompatible with strong oxidizing agents.
RUO – Research Use Only
Converts alcohols and phenols to their monosulfates under mild conditions: Ber., 57, 1045 (1924); J. Am. Chem. Soc., 74, 5212 (1952).
Reagent for the dehydration of oximes: Synthesis, 702 (1978).
Conditions have been described for the conversion of organolithiums in THF to sulfonic acids by insertion of SO3 into the C-Li bond: J. Org. Chem., 61, 1530 (1996):
Mans, D. J.; Ye, H.; Dunn, J. D.; Kolinski, R. E.; Long, D. S.; Phatak, N. L.; Ghasriani, H.; Buhse, L. F.; Kauffman, J. F.; Keire, D. A. Synthesis and detection of N-sulfonated oversulfated chondroitin sulfate in marketplace heparin. Anal. Biochem. 2015, 490, 52-54.
Jacquinet, J. C.; Lopin-Bon, C. Stereocontrolled preparation of biotinylated chondroitin sulfate E di-, tetra-, and hexasaccharide conjugates. Carbohydr. Res. 2015, 402, 35-43.
Reagent for the dehydration of oximes: Synthesis, 702 (1978).
Conditions have been described for the conversion of organolithiums in THF to sulfonic acids by insertion of SO3 into the C-Li bond: J. Org. Chem., 61, 1530 (1996):
Mans, D. J.; Ye, H.; Dunn, J. D.; Kolinski, R. E.; Long, D. S.; Phatak, N. L.; Ghasriani, H.; Buhse, L. F.; Kauffman, J. F.; Keire, D. A. Synthesis and detection of N-sulfonated oversulfated chondroitin sulfate in marketplace heparin. Anal. Biochem. 2015, 490, 52-54.
Jacquinet, J. C.; Lopin-Bon, C. Stereocontrolled preparation of biotinylated chondroitin sulfate E di-, tetra-, and hexasaccharide conjugates. Carbohydr. Res. 2015, 402, 35-43.