Search Thermo Fisher Scientific
Thermo Scientific Chemicals
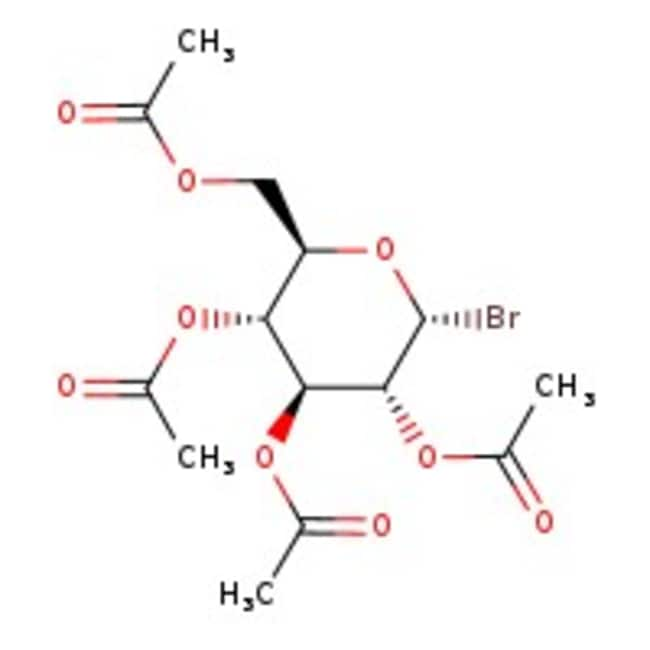
Acetobromo-alpha-D-glucose, 97%, stab. with 2% calcium carbonate, Thermo Scientific ChemicalsCatalog number L04151.18
also known as L04151-18
Price (USD)/ Each
299.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
352.00 Save 52.35 (15%)
-
Quantity:
50 g
Price (USD)/ Each
299.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
352.00 Save 52.35 (15%)
Acetobromo-alpha-D-glucose, 97%, stab. with 2% calcium carbonate, Thermo Scientific Chemicals
Catalog numberL04151.18
Price (USD)/ Each
299.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
352.00 Save 52.35 (15%)
-
Chemical Identifiers
CAS572-09-8
IUPAC Name[(2R,3R,4S,5R,6R)-3,4,5-tris(acetyloxy)-6-bromooxan-2-yl]methyl acetate
Molecular FormulaC14H19BrO9
InChI KeyCYAYKKUWALRRPA-HAGGZJDPNA-N
SMILESCC(=O)OC[C@H]1O[C@H](Br)[C@H](OC(C)=O)[C@@H](OC(C)=O)[C@@H]1OC(C)=O
View more
Specifications Specification Sheet
Specification Sheet
Formpowder
Assay (Titration ex Bromide)>96.0% (corrected for stabilser)
Optical Rotation+193 ? 7? ( (c=3 in chloroform)
Appearance (Color)White to pale cream or pale grey
Acetobromo-α-D-glucose acts as an intermediate in the preparation of beta-glucosides. It is also used as a possible poly(ethylene terephthalate) surface modification reagent to enhance its blood compatibility.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Acetobromo-α-D-glucose acts as an intermediate in the preparation of beta-glucosides. It is also used as a possible poly(ethylene terephthalate) surface modification reagent to enhance its blood compatibility.
Solubility
Soluble in ether, chloroform, ethanol, acetone, dichloromethane, ether, ethyl, methanol, and tetrahydrofuran. Insoluble in water.
Notes
Store in a cool place. Incompatible with strong oxidizing agents, acids, magnesium and aluminum.
Acetobromo-α-D-glucose acts as an intermediate in the preparation of beta-glucosides. It is also used as a possible poly(ethylene terephthalate) surface modification reagent to enhance its blood compatibility.
Solubility
Soluble in ether, chloroform, ethanol, acetone, dichloromethane, ether, ethyl, methanol, and tetrahydrofuran. Insoluble in water.
Notes
Store in a cool place. Incompatible with strong oxidizing agents, acids, magnesium and aluminum.
RUO – Research Use Only
Glycosyl isothiocyanates have been prepared in good yield by heating fully O-acetylated glycosyl bromides with KSCN in the absence of solvent. With acetobromo-ɑ-D-glucose, displacement was accompanied by stereochemical inversion: Synthesis, 1228 (1995).
Reaction with Bis(cyclopentadienyl)titanium dichloride, A11456 results in 1,2-elimination providing a convenient route to the glycal: J. Org. Chem., 60, 7055 (1995).
Forget, S. M.; Jee, A.; Smithen, D. A.; Jagdhane, R.; Anjum, S.; Beaton, S. A.; Palmer, D. R. J.; Syvitski, R. T.; Jakeman, D. L. Kinetic evaluation of glucose 1-phosphate analogues with a thymidylyltransferase using a continuous coupled enzyme assay. Org. Biomol. Chem. 2015, 13 (3), 866-875.
Xu, Y.; Wang, W.; Cai, Y.; Yang, X.; Wang, P. G.; Zhao, W. A convenient and efficient synthesis of glycals by zinc nanoparticles. RSC Adv. 2014, 4 (87), 46662-46665.
Reaction with Bis(cyclopentadienyl)titanium dichloride, A11456 results in 1,2-elimination providing a convenient route to the glycal: J. Org. Chem., 60, 7055 (1995).
Forget, S. M.; Jee, A.; Smithen, D. A.; Jagdhane, R.; Anjum, S.; Beaton, S. A.; Palmer, D. R. J.; Syvitski, R. T.; Jakeman, D. L. Kinetic evaluation of glucose 1-phosphate analogues with a thymidylyltransferase using a continuous coupled enzyme assay. Org. Biomol. Chem. 2015, 13 (3), 866-875.
Xu, Y.; Wang, W.; Cai, Y.; Yang, X.; Wang, P. G.; Zhao, W. A convenient and efficient synthesis of glycals by zinc nanoparticles. RSC Adv. 2014, 4 (87), 46662-46665.