Search Thermo Fisher Scientific
Thermo Scientific Chemicals
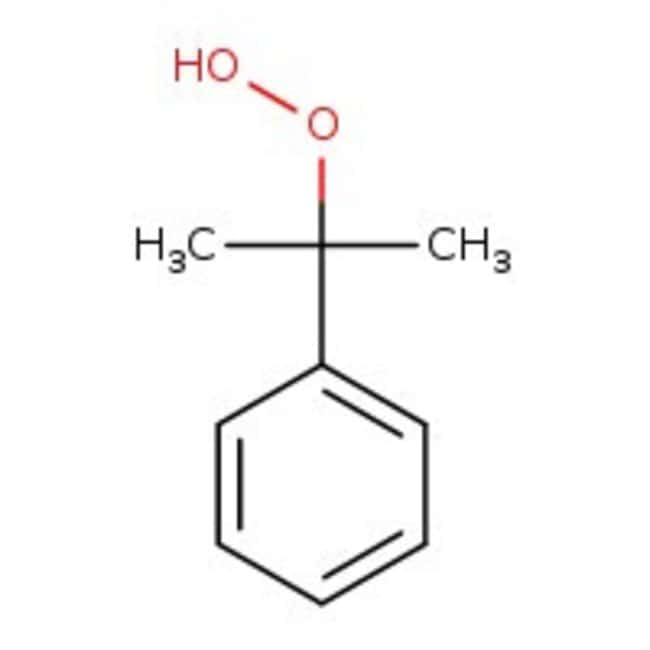
Cumyl hydroperoxide, tech. 80%, Thermo Scientific Chemicals
Catalog number L06866.36
also known as L06866-36
Price (USD)/ Each
85.65
Special Offer
Online exclusive
Ends: 31-Dec-2024
101.00 Save 15.35 (15%)
-
Quantity:
500 g
Price (USD)/ Each
85.65
Special Offer
Online exclusive
Ends: 31-Dec-2024
101.00 Save 15.35 (15%)
Cumyl hydroperoxide, tech. 80%, Thermo Scientific Chemicals
Catalog numberL06866.36
Price (USD)/ Each
85.65
Special Offer
Online exclusive
Ends: 31-Dec-2024
101.00 Save 15.35 (15%)
-
Chemical Identifiers
CAS80-15-9
IUPAC Name2-phenylpropane-2-peroxol
Molecular FormulaC9H12O2
InChI KeyYQHLDYVWEZKEOX-UHFFFAOYSA-N
SMILESCC(C)(OO)C1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Identification (FTIR)Conforms (U.K. specification)
FormLiquid
CommentPurchased in the U.K. and in other countries
Refractive Index1.5170-1.5250 @ 20°C
Appearance (Color)Clear colorless to pale yellow
View more
Cumene hydroperoxide is used in the preparation of polystyrene nanocapsules. It acts as a curing agent for polyester resins and as an oxidizer in organic chemical reactions. It serves as an initiator for radical polymerization especially for acrylate and methacrylate monomers. It also employed as an intermediate in the cumene process for developing phenol and acetone from benzene and propene. Further, it is used as an epoxidation reagent for allylic alcohols and fatty acid esters. In addition to this, it is also used to prepare methylstyrene, acetophenone and cumyl alcohol.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Cumene hydroperoxide is used in the preparation of polystyrene nanocapsules. It acts as a curing agent for polyester resins and as an oxidizer in organic chemical reactions. It serves as an initiator for radical polymerization especially for acrylate and methacrylate monomers. It also employed as an intermediate in the cumene process for developing phenol and acetone from benzene and propene. Further, it is used as an epoxidation reagent for allylic alcohols and fatty acid esters. In addition to this, it is also used to prepare methylstyrene, acetophenone and cumyl alcohol.
Solubility
Miscible with alcohol, acetone, ether, esters, hydrocarbons and chlorinated hydrocarbons. Slightly miscible with water.
Notes
Store in a cool place. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with powdered metals, organic materials, heavy metal salts, metal salts, combustible materials, acids, alkalis, reducing agents, rust, charcoal, amines, copper, lead, cobalt and cobalt oxides.
Cumene hydroperoxide is used in the preparation of polystyrene nanocapsules. It acts as a curing agent for polyester resins and as an oxidizer in organic chemical reactions. It serves as an initiator for radical polymerization especially for acrylate and methacrylate monomers. It also employed as an intermediate in the cumene process for developing phenol and acetone from benzene and propene. Further, it is used as an epoxidation reagent for allylic alcohols and fatty acid esters. In addition to this, it is also used to prepare methylstyrene, acetophenone and cumyl alcohol.
Solubility
Miscible with alcohol, acetone, ether, esters, hydrocarbons and chlorinated hydrocarbons. Slightly miscible with water.
Notes
Store in a cool place. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with powdered metals, organic materials, heavy metal salts, metal salts, combustible materials, acids, alkalis, reducing agents, rust, charcoal, amines, copper, lead, cobalt and cobalt oxides.
WARNING: Cancer – www.P65Warnings.ca.gov
RUO – Research Use Only
General References:
- Alternative to tert-Butyl hydroperoxide, A13926, in certain catalytic peroxygenation reactions. For example, has been found to give better enantiomeric excesses in the Sharpless-type asymmetric oxidation of sulfides to sulfoxides in the presence of Ti(O-i-Pr)4 and diethyl L-tartrate: Tetrahedron, 43, 5135 (1987); Org. Synth. Coll., 8, 464 (1993); Synlett, 404 (1996).
- Pardillo-Díaz, R.; Carrascal, L.; Ayala, A.; Nunez-Abades, P. Oxidative stress induced by cumene hydroperoxide evokes changes in neuronal excitability of rat motor cortex neurons. Neuroscience 2015, 289, 85-98.
- Liu, J.; Ni, X.; Hu, Y. Influence of nitridation on the catalytic performance of Ti-MCM-41 for the epoxidation of propene by cumene hydroperoxide. Reac Kinet Mech Cat 2015, 114 (2), 685-695.