Search Thermo Fisher Scientific
Thermo Scientific Chemicals
Manganese(IV) oxide activated, tech. 90%, Thermo Scientific ChemicalsCatalog number L13253.36
also known as L13253-36
Price (USD)/ Each
86.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
102.00 Save 15.35 (15%)
-
Quantity:
500 g
Price (USD)/ Each
86.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
102.00 Save 15.35 (15%)
Manganese(IV) oxide activated, tech. 90%, Thermo Scientific Chemicals
Catalog numberL13253.36
Price (USD)/ Each
86.65
Special Offer
Online exclusive
Ends: 15-Oct-2024
102.00 Save 15.35 (15%)
-
Chemical Identifiers
CAS1313-13-9
IUPAC Namedioxomanganese
Molecular FormulaMnO2
InChI KeyNUJOXMJBOLGQSY-UHFFFAOYSA-N
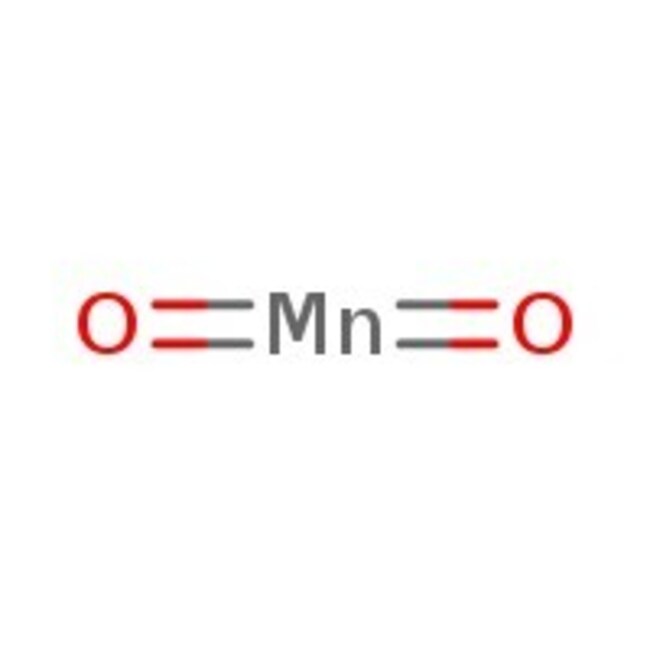
SMILESO=[Mn]=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Black
Assay from Suppliers CofA≥88.0% (≥55.6% Mn)
FormPowder
Manganese(IV) oxide is suitable for use in batteries. It is widely used for the selective oxidation of allylic and benzylic alcohols to aldehydes or ketones, as a co-oxidant in combination with 1,4-benzoquinone and in the allylic oxidation of olefins catalyzed by Palladium(II) acetate. It acts a reagent for oxidations. It is the source of manganese and all its compounds, largely used in manufacturing of manganese steel, for making amethyst glass, decolorizing glass, painting on porcelain, faience and majolica. The precipitate is used in electrotechnics, pigments, browning gun barrels, drier for paints and varnishes, printing and dyeing textiles.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Manganese(IV) oxide is suitable for use in batteries. It is widely used for the selective oxidation of allylic and benzylic alcohols to aldehydes or ketones, as a co-oxidant in combination with 1,4-benzoquinone and in the allylic oxidation of olefins catalyzed by Palladium(II) acetate. It acts a reagent for oxidations. It is the source of manganese and all its compounds, largely used in manufacturing of manganese steel, for making amethyst glass, decolorizing glass, painting on porcelain, faience and majolica. The precipitate is used in electrotechnics, pigments, browning gun barrels, drier for paints and varnishes, printing and dyeing textiles.
Solubility
Insoluble in water.
Notes
Store at room temperature. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with strong oxidizing agents.
Manganese(IV) oxide is suitable for use in batteries. It is widely used for the selective oxidation of allylic and benzylic alcohols to aldehydes or ketones, as a co-oxidant in combination with 1,4-benzoquinone and in the allylic oxidation of olefins catalyzed by Palladium(II) acetate. It acts a reagent for oxidations. It is the source of manganese and all its compounds, largely used in manufacturing of manganese steel, for making amethyst glass, decolorizing glass, painting on porcelain, faience and majolica. The precipitate is used in electrotechnics, pigments, browning gun barrels, drier for paints and varnishes, printing and dyeing textiles.
Solubility
Insoluble in water.
Notes
Store at room temperature. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with strong oxidizing agents.
RUO – Research Use Only
Jay A. Young. Manganese(IV) Oxide. J. Chem. Educ. 2001, 78 (10), 1327.
Jose Alberto Cerri.; Edson Roberto Leite.; Douglas Gouvêa.; Elson Longo.; Jose Arana Varela. Effect of Cobalt(II) Oxide and Manganese(IV) Oxide on Sintering of Tin(IV) Oxide. Journal of the American Ceramic Society. 1996, 79 (3), 799-804.
Mild oxidizing agent. For reviews, see: Synthesis, 65, 133 (1976); J. Prakt. Chem./ Chem. Ztg., 337, 75 (1995). See also M. Hudlicky, Oxidations in Organic Chemistry, ACS Monograph 186, Washington DC (1990).
Widely used for the selective oxidation of allylic and benzylic alcohols to aldehydes or ketones. See, e.g.: Org. Synth. Coll., 7, 102 (1990); 9, 136 (1998).
For use as a co-oxidant, in combination with 1,4-benzoquinone, in the allylic oxidation of olefins, catalyzed by Palladium(II) acetate, 10516, see: Org. Synth. Coll., 8, 5, 137 (1993).
Jose Alberto Cerri.; Edson Roberto Leite.; Douglas Gouvêa.; Elson Longo.; Jose Arana Varela. Effect of Cobalt(II) Oxide and Manganese(IV) Oxide on Sintering of Tin(IV) Oxide. Journal of the American Ceramic Society. 1996, 79 (3), 799-804.
Mild oxidizing agent. For reviews, see: Synthesis, 65, 133 (1976); J. Prakt. Chem./ Chem. Ztg., 337, 75 (1995). See also M. Hudlicky, Oxidations in Organic Chemistry, ACS Monograph 186, Washington DC (1990).
Widely used for the selective oxidation of allylic and benzylic alcohols to aldehydes or ketones. See, e.g.: Org. Synth. Coll., 7, 102 (1990); 9, 136 (1998).
For use as a co-oxidant, in combination with 1,4-benzoquinone, in the allylic oxidation of olefins, catalyzed by Palladium(II) acetate, 10516, see: Org. Synth. Coll., 8, 5, 137 (1993).