Search Thermo Fisher Scientific
Thermo Scientific Chemicals
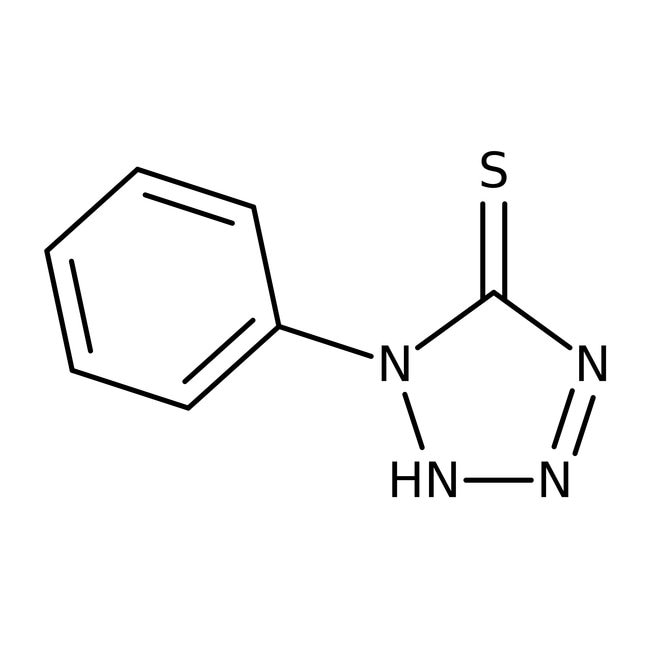
1-Phenyl-1H-tetrazole-5-thiol, 99%, Thermo Scientific ChemicalsCatalog number A12656.22
also known as A12656-22
Price (EUR)/ Each
112,50
Online exclusive
125.00 Save 12,50 (10%)
-
Quantity:
100 g
Price (EUR)/ Each
112,50
Online exclusive
125.00 Save 12,50 (10%)
1-Phenyl-1H-tetrazole-5-thiol, 99%, Thermo Scientific Chemicals
Catalog numberA12656.22
Price (EUR)/ Each
112,50
Online exclusive
125.00 Save 12,50 (10%)
-
Chemical Identifiers
CAS86-93-1
IUPAC Name1-phenyl-2,5-dihydro-1H-1,2,3,4-tetrazole-5-thione
Molecular FormulaC7H6N4S
InChI KeyGGZHVNZHFYCSEV-UHFFFAOYSA-N
SMILESS=C1N=NNN1C1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to pale cream
FormCrystals or powder or crystalline powder
Assay (HPLC)≥98.5%
Identification (FTIR)Conforms
1-Phenyl-1H-tetrazole-5-thiol is an effective inhibitor of aluminum corrosion in 1M HCl solution. 1-Phenyl-1H-tetrazole-5-thiol was used in the synthesis of oxacyclic building blocks via highly stereoselective radical cyclization and olefin metathesis reactions. It was also used in the synthesis of metalated tetradecyl sulfone. It is also used in the spectrophotometric determination of Pt and Bi.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1-Phenyl-1H-tetrazole-5-thiol is an effective inhibitor of aluminum corrosion in 1M HCl solution. 1-Phenyl-1H-tetrazole-5-thiol was used in the synthesis of oxacyclic building blocks via highly stereoselective radical cyclization and olefin metathesis reactions. It was also used in the synthesis of metalated tetradecyl sulfone. It is also used in the spectrophotometric determination of Pt and Bi.
Solubility
Soluble in 5% ethanol, water (partly), acetone, chloroform, and methanol.
Notes
Air Sensitive. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Stable under recommended storage conditions. Keep away from oxidizing agents.
1-Phenyl-1H-tetrazole-5-thiol is an effective inhibitor of aluminum corrosion in 1M HCl solution. 1-Phenyl-1H-tetrazole-5-thiol was used in the synthesis of oxacyclic building blocks via highly stereoselective radical cyclization and olefin metathesis reactions. It was also used in the synthesis of metalated tetradecyl sulfone. It is also used in the spectrophotometric determination of Pt and Bi.
Solubility
Soluble in 5% ethanol, water (partly), acetone, chloroform, and methanol.
Notes
Air Sensitive. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Stable under recommended storage conditions. Keep away from oxidizing agents.
RUO – Research Use Only
General References:
- R Cea-Olivares.; OJ Sandoval.; G Espinosa-Perez. New organotin(IV) derivatives of 1-phenyl-1H-tetrazole-5-thiol. Crystal and molecular structure of 1∞[Bz3SnSCN4Ph], a polymer built through secondary intermolecular Sn⋯N bonds. Polyhedron. 2008, 13, (19), 2809-2818.
- C Ma.; J Sun. Hydrogen bonding and π-π stacking interaction in the coordination of sulfur and nitrogen donor heterocycle to organotin(IV): syntheses and crystal structures of di- and triorganotin(IV) derivatives with 1-(4-hydroxyphenyl)-1H-tetrazole-5-thiol. Polyhedron. 2004, 23, (9), 2809-2818.
- In combination with t-butyl isocyanide, promotes the lactonization of long-chain hydroxy acids: Angew. Chem. Int. Ed., 20, 771 (1981).