Search Thermo Fisher Scientific
Clinical Specimen Collection
Clinical specimen collection are specially designed supplies used to collect clinical samples, which include saliva, urine, and stool. These products are available in various sizes and materials.
Microbiological Specimen Transport and Collection Kits (48)

General Clinical Specimen Containers (35)

Stool Specimen Collection (28)
Urine Collection (6)

Specimen Labelers and Accessories (2)
Products (119)
Learn More (50)
Documents & Support
(124)119 Products
Filter
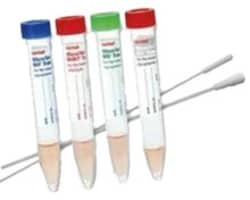
MicroTest™ M4RT 3mL Flocked Swab Kit Thermo Scientific™
Maintain organism viability and counts for verification studies, quality control procedures, and repeat testing of viruses chlamydiae, mycoplasma and ureaplasma with quality media, proven collection and transport tools, and safe, reliable packaging.
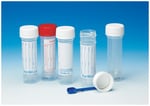
Samco™ Clicktainer™ Vials and Specimen Containers Thermo Scientific™
Providing sample protection and user safety with the Thermo Scientific™ Samco™ Clicktainer™ Vials and Specimen container, equipped with an audible multi-click seal. Help enable improved patient diagnosis with a leak tight seal, easy to grip surfaces and straight sided design for reliable and safe...
Nunc™ Urine Sample Kits Thermo Scientific™
Thermo Scientific™ Nunc™ Urine Sample Kits. Nunc Labware Products are made from high purity resins and molded using our state-of-the-art processes. Kits are sterile and easy to use for collection and transportation of urine samples specifically for in vitro diagnostic for patients who need...
Sterilin™ Triple Bagged Polystyrene Containers Thermo Scientific™
Use Thermo Scientific™ Sterilin™ Triple Bagged Polystyrene Containers in hospital rooms and other such sterile areas. NOT AVAILABLE IN AMERICA
MicroTest™ Universal Kits Thermo Scientific™
Maintain organism viability and counts for verification studies, quality control procedures, and repeat testing of viruses, chlamydiae, mycoplasma and ureaplasma with quality media, proven collection and transport tools, and safe, reliable packaging.
Nunc™ Universal and Transport Containers with Caps Thermo Scientific™
Safely store and transport blood, urine, fecal and other specimens using Thermo Scientific™ Nunc™ Universal and Transport Containers with Caps. These containers are designed with inner and outer containers of high mechanical strength.
Nunc™ Containers Thermo Scientific™
Store samples safely using Thermo Scientific™ Nunc™ Containers. Nunc Labware Products are made from high purity resins and molded using our state-of-the-art processes. Plastic labware is a safer alternative to glass without sacrificing accuracy.
MicroTest™ Universal Plus Kits Thermo Scientific™
Maintain organism viability and counts for verification studies, quality control procedures, and repeat testing of viruses chlamydiae, mycoplasma and ureaplasma with quality media, proven collection and transport tools, and safe, reliable packaging.
Sterilin™ Polystyrene Containers, 60mL to 250mL Thermo Scientific™
Contain samples in Thermo Scientific™ Sterilin™ Polystyrene Containers with 60mL to 250mL capacities. Available with either plastic or metal flow seal caps. NOT AVAILABLE IN AMERICA
Richard-Allan Scientific™ Bio-Tite™ Specimen Containers,Formalin Warning Labels, Large Thermo Scientific™
Transport, store and retrieve pathology specimens in a safe and cost effective manner with the Thermo Scientific™ Richard-Allan Scientific™ Specimen Transportation System (STS™). This system includes containers pre-filled with the highest quality 10% Neutral Buffered Formalin (NBF) and Zinc...
Samco™ Narrow Mouth Bio-Tite™ 40mL (1 oz.) 48mm Specimen Containers Thermo Scientific™
Prevent bio-hazardous spills and conserve storage space with stackable Thermo Scientific™ Samco™ Narrow-Mouth Bio-Tite™ 40mL (1 oz.) 48mm Specimen Containers. These shatter-resistant containers feature a proprietary, all plastic, leak-resistant seal.
Samco™ Wide-Mouth Bio-Tite™ 90mL (3 oz.) 53mm Specimen Containers Thermo Scientific™
Prevent bio-hazardous spills with the proprietary, all plastic, leak-resistant seal of Thermo Scientific™ Samco™ Wide-Mouth Bio-Tite™ 90mL (3 oz.) 53mm Specimen Containers. Wide-mouth containers are ideally suited for urine collection from women during pregnancy and for older or overweight patients.
Samco™ Narrow Mouth Bio-Tite™ 60mL (2 oz.) 48mm Specimen Containers Thermo Scientific™
Prevent bio-hazardous spills and conserve storage space with stackable Thermo Scientific™ Samco™ Narrow-Mouth Bio-Tite™ 60mL (2 oz.) 48mm Specimen Containers. These shatter-resistant containers feature a proprietary, all plastic, leak-resistant seal.
Sterilin™ Polypropylene 30mL Universal Containers Thermo Scientific™
Secure excellent temperature and chemical resistance with these 95kPa compliant 30mL leak-resistant Thermo Scientific™ Sterilin™ Polypropylene 30mL Universal Containers. NOT AVAILABLE IN AMERICA
Sterilin™ Polystyrene 30mL Universal Containers Thermo Scientific™
Ensure excellent sample containment using Thermo Scientific™ Sterilin Polystyrene 30mL Universal Containers, featuring a traditional flow seal with 1-1/2 turn cap. NOT AVAILABLE IN AMERICA
Learn More (50)
View all
Available in a wide range of formulations, Thermo Scientific™ Specimen Collection and Transport Solutions enable simple and safe collection and transport of all specimen types. The majority of our products are designed for automation — further simplifying your workflow and eliminating the need for...
At Thermo Fisher Scientific Microbiology, we have remained focused and flexible to ramp up production and deliver the products that you rely on. This is all to meet your testing demands during the coronavirus pandemic and beyond.
Documents & Support (124)
View all
Interim Guidelines for Collecting, Handling, and Testing Clinical Specimens
SmartNote: Coronavirus: CDC Interim Guidelines for Collecting, Handling, and Testing Clinical Specimens